Context
Dehydroepiandrosterone (DHEA) and DHEA sulfate (DHEAS) are the major circulating adrenal steroids and substrates for peripheral sex hormone biosynthesis. In Addison’s disease, glucocorticoid and mineralocorticoid deficiencies require lifelong replacement, but the associated near-total failure of DHEA synthesis is not typically corrected.
Objective and design
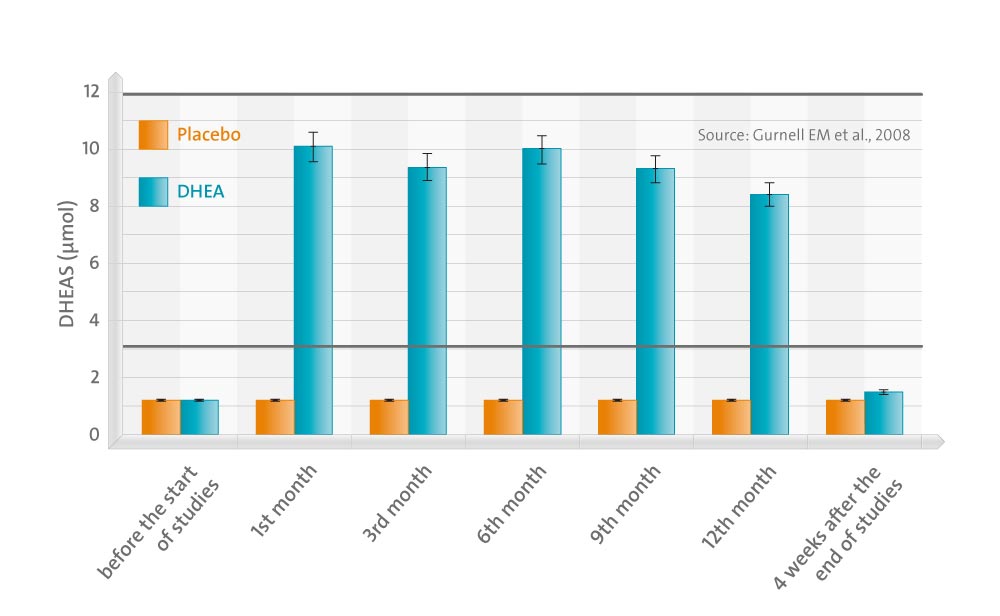
In a double-blind trial, we randomized 106 subjects (44 males, 62 females) with Addison’s disease to receive either 50 mg daily of micronized DHEA or placebo orally for 12 months to evaluate its longer-term effects on bone mineral density, body composition, and cognitive function together with well-being and fatigue.
Results
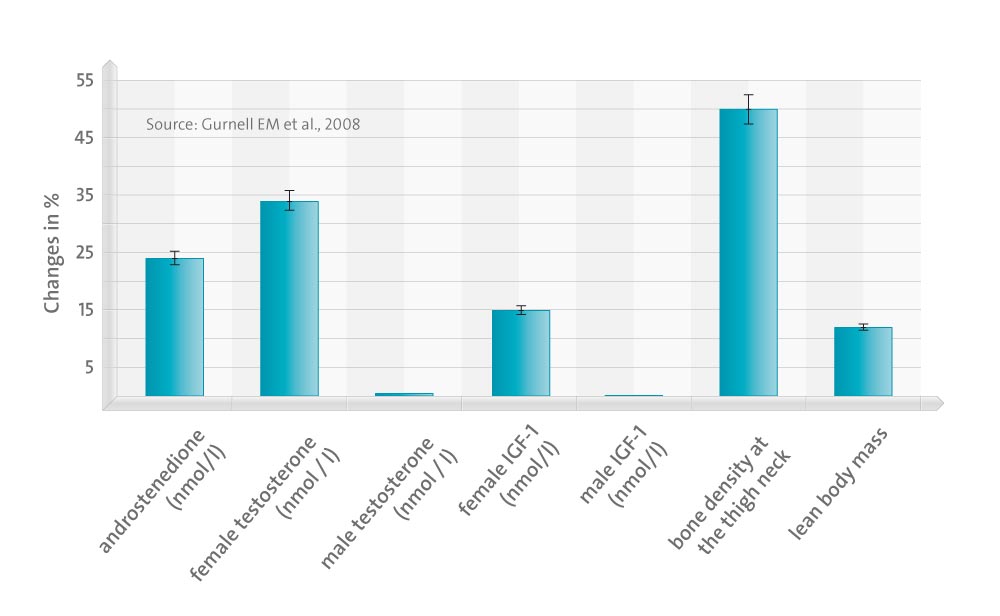
Circulating DHEAS and androstenedione rose significantly in both sexes, with testosterone increasing to low normal levels only in females. DHEA reversed ongoing loss of bone mineral density at the femoral neck (P < 0.05) but not at other sites; DHEA enhanced total body (P = 0.02) and truncal (P = 0.017) lean mass significantly with no change in fat mass. At baseline, subscales of psychological well-being in questionnaires (Short Form-36, General Health Questionnaire-30), were significantly worse in Addison’s patients vs. control populations (P < 0.001), and one subscale of SF-36 improved significantly (P = 0.004) after DHEA treatment. There was no significant benefit of DHEA treatment on fatigue or cognitive or sexual function. Supraphysiological DHEAS levels were achieved in some older females who experienced mild androgenic side effects.
Conclusion
Although further long-term studies of DHEA therapy, with dosage adjustment, are desirable, our results support some beneficial effects of prolonged DHEA treatment in Addison’s disease.